Urea
Praèn
| Urea | |
|---|---|
| |
| Umum | |
| Jeneng internasional | Diaminomethanal |
| Jeneng liya | carbamide |
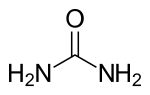
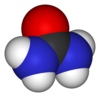
| Rumus Molekul | (NH2)2CO |
| Rumus bangun | NC(=O)N |
| Massa atom | 60.07 g/mol |
| Penampakan | padhetan awarna putih tanpa ambu |
| CAS number | [57-13-6] |
| Properties | |
| Berat Jinis and phase | 1.33•103 kg/m3 [1], solid |
| Kelarutan sajeroning banyu | 108 g/100 ml (20 °C)167 g/100 ml (40 °C)251 g/100 ml (60 °C)400 g/100 ml (80 °C)733 g/100 ml (100 °C) |
| Titik lebur | 132.7 °C (406 K)decomposes |
| Titik didih | n.a. |
| Keasaman (pKa) | 0.18 |
| Kebasaan (pKb) | 13.82 |
| Chiral rotation [α]D | Not chiral |
| Kakenthelan | ? cP at ? °C |
| Kelembaban relatif kritis | 81% (20 °C)73% (30 °C) |
| Heat of solution in water | -57,8 cal/g (endothermic) |
| 46,6 %N | |
| Struktur | |
| Molecular shape | ? |
| Coordination geometry | trigonal planar |
| Crystal structure | ? |
| Dipole moment | ? D |
| Hazards | |
| MSDS | J.T. Baker |
| Main hazards | ? |
| Titik nyala | ? °C |
| R/S statement | R: ? S: ? |
| RTECS number | ? |
| NFPA 704 | Cithakan:NFPA 704 estimated |
| Informasi tambahan | |
| Structure & properties | n, εr, etc. |
| Thermodynamic data | Phase behaviourSolid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other anions | ? |
| Other cations | ? |
| Related ? | biurettriuretthiourea |
| Related compounds | ? |
| Yèn ora ana wedharan tambahan, materi ana ing kondisi standar (at 26 °C, 1 atm)Infobox disclaimer and references | |
Urea iku siji senyawa organik kang dumadi saka unsur karbon, hidrogen, oksigèn lan nitrogen kanthi rumus CON2H4 utawa (NH2)2CO. Urea uga dikenal kanthi jeneng carbamide kang mligi dipigunakaké ing kawasan Éropah. Jeneng liya kang uga kerep dianggo ya iku carbamide resin, isourea, carbonyl diamide lan carbonyldiamine. Senyawa iki wujud senyawa organik sintesis pisanan kang kasil digawé saka senyawa anorganik, kang pungkasané ngruntuhaké konsèp vitalisme.
Panemuan
[besut | besut sumber]Urea tinemu déning Hilaire Roulle taun 1773. Senyawa iki minangka senyawa organik pisanan kang kasil disintesis saka senyawa anorganik.
Rujukan
[besut | besut sumber]
| Artikel kimia iki minangka artikel rintisan. Kowé bisa ngéwangi Wikipédia ngembangaké. |

